Environmental swabbing remains one of the most critical yet frequently misunderstood components of food safety and quality systems. The recurring questions from QA managers and food safety professionals revolve around a practical concern: Why doesn’t our environmental data always reflect what we see on the floor? When results fluctuate without clear cause, confidence in the Environmental Monitoring Program begins to erode.
An Environmental Monitoring Program (EMP) is fundamentally a system of evidence. Its strength depends entirely on how samples are collected, handled, and preserved before analysis. A poorly selected swab, inappropriate timing, or a break in temperature control can distort results long before they reach the laboratory. The consequences include misleading negatives, inefficient corrective actions, and exposure during regulatory audits.
This article lays out the seven most common technical failures in environmental swabbing, explains the scientific reason each one breaks your data, and gives concise, implementable fixes.
1. Selecting Non-Validated Swabs or Incorrect Neutralizers for Environmental Swabbing
Why it breaks your program:
Many basic cotton or dry swabs do not neutralize residual sanitizers. When a swab absorbs sanitizer residues on a surface, that sanitizer can continue to inactivate organisms on the swab during transport or enrichment, producing false negatives. Different sanitizers (chlorine, quats, peracetic acid) require different neutralizing chemistries; a one-size swab can fail against multiple chemistries.
Technical fix (stepwise):
- Specify swabs/sponges that contain validated neutralizing formulations (e.g., Dey-Engley or equivalent wide-spectrum neutralizers) and confirm vendor validation data for the sanitizers used onsite.
- Maintain a swab inventory matrix mapping each sanitizer to acceptable neutralizers and approved swab SKUs.
- Run an internal verification: inoculate a known surface, apply your sanitizer at the working concentration, then sample with your chosen swab and verify recovery against a control. Record results in your method verification file.
2. Ignoring Risk-Based Sampling Zones in Environmental Monitoring
Why it breaks your program:
Pathogens establish in protected, moist niches like drains, gaskets, and hollow rollers where routine cleaning may not reach. Sampling only convenient flat surfaces creates a highly biased and falsely reassuring dataset. Regulatory guidance and industry practice emphasize targeted, risk-based zoning.
Technical fix (stepwise):
- Build a risk map that classifies zones by contact risk, moisture, and cleanability. Tag each zone with the appropriate test panel (e.g., Listeria genus in wet niches).
- Use environmental mapping tools and rotating deep-sampling rounds to periodically target “low-use” niches. Document rationale for zone selection so your sampling plan is defensible during audits.
3. Non-Standardized Environmental Swabbing Techniques
Why it breaks your program:
Surface recovery is a function of contact area, wiping pattern, pressure, and number of passes. Inconsistent technique increases variance and makes trend analysis meaningless. Standards recommend defined areas (commonly 100 cm²) and repeatable patterns for comparability.
Technical fix (stepwise):
- Standardize a sampling template (e.g., 100 cm²) and a repeatable pattern (Z or S). Use templates (stencils) to mark the area for each sampler.
- Train with competency checks: have samplers collect replicate swabs on spiked surfaces and compare recovery to establish acceptable inter-operator variance.
- Lock technique into your SOP and require periodic retraining and sign-off.
4. Inadequate Temperature Control During Environmental Sample Transport
Why it breaks your program:
Environmental samples are sensitive: freezing can reduce viability of some bacteria, while elevated temperatures permit overgrowth. Both outcomes distort qualitative and quantitative results. Maintaining validated 2–8°C transport conditions is a widely accepted practice to preserve sample integrity during transit.
Technical fix (stepwise):
- Use validated insulated containers with appropriate coolants (ice packs or gel packs) and temperature loggers or time–temperature indicators for each transport batch.
- Define an acceptance window (e.g., receipt within X hours at 2–8°C) and a breach protocol that documents actions taken if the cold chain is broken.
- Train shipping staff on packing order (samples surrounded by coolant, not touching frozen packs) and lab receiving procedures.
5. Dilution and Cross-Contamination Risks from Multi-Pathogen Swab Testing
Why it breaks your program:
Dividing a single swab for different pathogen assays dilutes the organism load available to each test and increases cross-contamination risk between assays. Effective pathogen monitoring depends on high analytical sensitivity, which is reduced when samples are divided. Many authorities do not recommend splitting swabs for routine pathogen detection.
Technical fix (stepwise):
- Allocate dedicated swabs for each target pathogen or validated dual-side devices specifically approved by your lab method. Avoid splitting at all costs for routine pathogen surveillance.
- Cost-driven compositing should remain a screening tool only, supported by documented risk assessment, and never substitute for validated pathogen-specific testing.
6. Improper Environmental Monitoring Scheduling
Why it breaks your program:
While post-clean sampling validates sanitation effectiveness, it fails to measure contamination introduced during production. An effective EMP must assess both sanitation verification and in-process risk because organisms can accumulate during production or be introduced by personnel or equipment.
Technical fix (stepwise):
- Implement a mixed timepoint approach: pre-op (sanitation verification), in-process (mid-shift or peak production), and post-op (sanitation re-check) as appropriate to product risk.
- Use trend analysis to decide if additional targeted mid-shift or post-shift sampling is required for hotspots.
7. Non-Adherence to Clean-to-Dirty Sampling Protocols
Why it breaks your program:
Sampling from dirty to clean without changing gloves or tools can transfer contaminants and create false positives in low-risk areas, obstructing root-cause work and increasing corrective costs. Simple workflow discipline prevents this.
Technical fix (stepwise):
- Embed a clean-to-dirty sampling sequence in the SOP and sampling sheets. Require glove changes or fresh sleeves between zones and document the sequence of each event.
- Audit adherence through periodic observation and include sequence checks in your corrective action investigations.
Building a Sustainable and Compliant Environmental Monitoring Program (EMP)
Technical controls in an Environmental Monitoring Program (EMP) are essential but without governance, they don’t hold up under audit scrutiny. Sustainable environmental monitoring requires documented method verification, sampler competency records, calibrated temperature logs, and a traceable chain-of-custody system to transform routine swabbing into defensible microbiological evidence.
Aligning SOPs with globally recognized guidance such as ISO 18593 from the International Organization for Standardization ensures consistency, credibility, and regulatory readiness.
When governance frameworks, standardized procedures, and documented controls are fully embedded into your Environmental Monitoring Program, environmental swabbing evolves into a strategic risk management tool. However, maintaining this level of consistency manually can be resource-intensive and vulnerable to human error.
This is where digital integration and structured program oversight become critical.
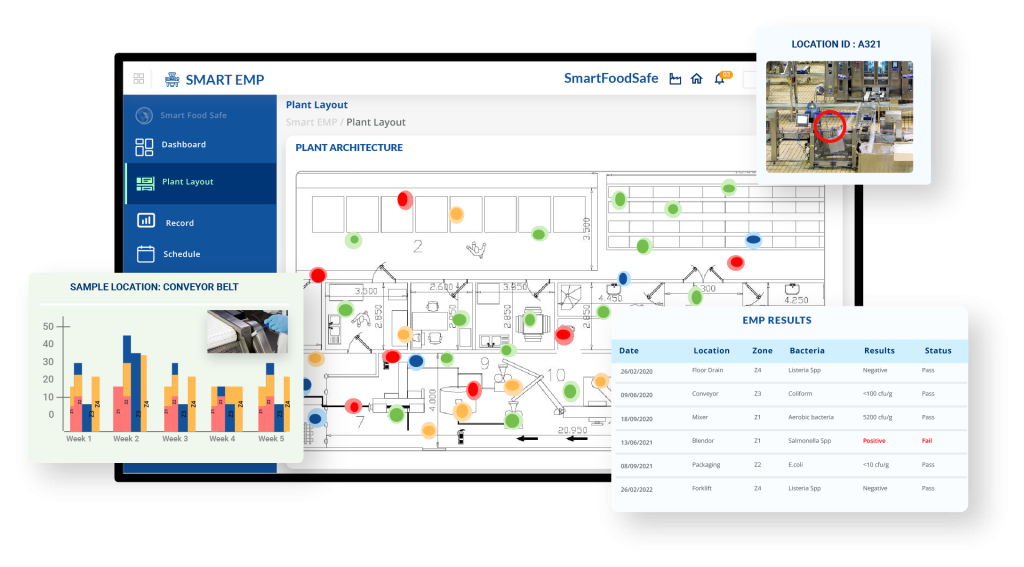
How Smart EMP Software Strengthens Environmental Monitoring Compliance and Reduces Risk
Smart EMP strengthens environmental monitoring by converting vulnerable manual processes into controlled, standardized workflows. It enforces zone-based protocol selection, maintains validated swab-to-sanitizer compatibility records, schedules both pre-operational and in-process sampling, and digitally captures chain-of-custody and cold-chain data. Sampler competency records and corrective actions are centralized within a single, audit-ready platform.
By embedding procedural requirements into system controls, Smart EMP minimizes variability, improves traceability, and transforms environmental swabbing data into reliable, decision-ready intelligence.
If you would like a professionally structured Environmental Swabbing SOP template, a 100 cm² sampling guide, or a demonstration of how Smart EMP strengthens environmental monitoring compliance and traceability, request a Demo today.
Explore our related resources to further strengthen your Environmental Monitoring Program
Environmental Monitoring Program (EMP) Best Practices: Strategic Guide to Effective Sampling
The Importance of Scheduling in Environmental Monitoring Program Sampling
Why Drains Matter So Much in Listeria Prevention for Ready-to-Eat Foods